News
Public Procurement: Medicines procured under four government programs are delivered to the regions
Medicines procured for the state budget funds under four government programs are currently delivered to the regions. These medicines will be available in health facilities in the nearest future.
Substitution Maintenance Therapy
- Buprenorphine hydrochloride tablets* – 9 806 packs of 4 mg tablets; 792 packs of 2 ml tablets;
- Methadone ZN tablets* – 2 920 packs of 5 mg tablets; 3 433 packs of 10 mg tablets; 20 400 packages of 25 mg tablets.
Child Nanism (Dwarfism)
- Growtropin* – 400 vials
Oncohematology
- Vancomycin-Vocate* – 51 packs
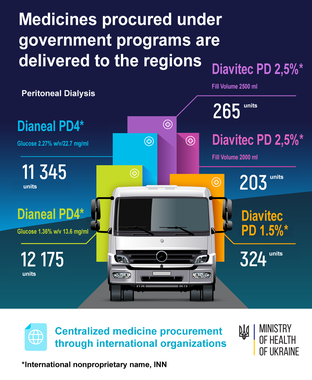
Peritoneal Dialysis
- Dianeal PD4* Glucose 2.27% w/v / 22.7 mg/ml – 11 345 units;
- Dianeal PD4* Glucose 1.36% w/v 13.6 mg/ml – 12 175 units;
- Diavitec PD 2,5%* Fill Volume 2500 ml – 265 units;
- Diavitec PD 2.5%* Fill Volume 2000 ml – 203 units;
- Diavitec PD 1.5%* – 324 units.



The Ministry of Health of Ukraine procures medicines and healthcare products for the funds allocated from the state budget – the amount annually approved by the Verkhovna Rada of Ukraine (UAH 3.4 billion in 2016, UAH 5.9 billion in 2017, and UAH 5.9 billion in 2018). On average, about 45% of the total demand for medicine procurement is covered by the national sources – funds allocated from the state budget.
Medicine procurement through specialized international organizations allows negotiating on prices, and therefore, increasing the purchase volume.
In case when specific medicines are available in stock at hospital, but a patient does not receive it free of charge for any reason, this patient should submit a written notice to the chief physician of this particular health facility or to the local administration / health department, indicating diagnosis with the relevant testing or screening results, and the prescribed medicines.
Order of the Ministry of Health of Ukraine No. 509 stipulates that all health facilities and institutions are obliged to publish information on availability in stock of medicines and healthcare products.
Detailed information about medicine procurement is available in Ukrainian: http://moz.gov.ua/zakupivli-likiv



